Archive for the ‘Stem Cell Therapy’ Category
Central Cord Syndrome
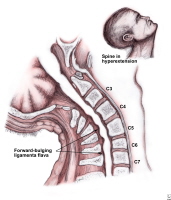
Central Cord Syndrome (CCS) is an incomplete injury to the cervical cord resulting in more extensive motor weakness in the upper extremities than the lower extremities. The mechanism of injury occurs from a hyperextension injury with pre-existent osteophytic (abnormal bony outgrowth) spurs, without damage to the vertebral column.
Mechanism and Causes of Injury
CCS occurs typically in patients with hyperextension injuries where the spinal cord is squeezed or pinched between anterior cervical spondylotic bone spurs and the posterior intraspinal canal ligament, the ligamentum flavum. The ligamentum flavum is a strong ligament that connects the laminae of the vertebrae. It serves to protect the neural elements and the spinal cord and stabilize the spine so that excessive motion between the vertebral bodies does not occur.
The injury occurs as a result of anterior and posterior compression of the spinal cord, leading to edema, hemorrhage or ischemia to the central portion of the spinal cord. The site of most injuries is in the mid-to-lower cervical cord. Due to the anatomical lamination of the corticospinal tract with the arm fibers medially, and the leg fibers laterally, the arms are affected more so than the legs, resulting in a disproportionate motor impairment.
Symptoms
Patients are typically left with motor weakness of the upper extremities and lesser involvement of the lower extremities. A varying degree of sensory loss below the level of the lesion and bladder symptoms (urinary retention) may both occur.
Incidence
This syndrome more commonly affects patients age 50 and older who have sustained a cervical hyperextension injury.
CCS may occur in patients of any age and is seen in athletes who present with not only hyperextension injuries to their neck but associated ruptured disc(s) with anterior cord compression.
CCS affects males more frequently than females.
Diagnosis
Evaluation of the patient includes a complete history, a thorough neurological exam, MRI and CT of the cervical spine, and cervical spine x-rays including supervised flexion and extension x-rays.
- Magnetic resonance imaging (MRI): A diagnostic test that produces three-dimensional images of body structures using powerful magnets and computer technology; can show direct evidence of spinal cord impingement from bone, disc, or hematoma.
- Computed tomography scan (CT or CAT scan): A diagnostic image created after a computer reads x-rays; can show the shape and size of the spinal canal, its contents, and the structures around it.
- X-ray: Application of radiation to produce a film or picture of a part of the body can show the structure of the vertebrae and the outline of the joints. X-rays of the spine delineate fractures and dislocations, as well as the degree and extent of spondylitic changes. Flexion/extension views assist in evaluation of ligamentous stability.
Surgical Treatment
Acute surgical intervention is not usually necessary unless there is significant cord compression. Prior to the CT-MRI era, surgical intervention was thought to be more harmful because of the risk of injuring a swollen cervical cord and worsening the deficit. However, with advanced imaging technology such as CT and MRI, patients with compression of the spinal cord secondary to traumatic herniated discs and other lesions can be quickly diagnosed and surgically decompressed. In cases with anterior bony ridges and spinal canal narrowing secondary to ligamentous thickening and or stenosis, acute surgery is usually not performed until the patient has made maximum recovery. Reassessment at that time may lead to surgery depending on the underlying cause. If there is significant motor weakness after a period of recovery, or neurological deterioration or spinal instability, then surgical intervention may be considered.
Nonsurgical Treatment
Nonsurgical treatment consists of immobilization of the neck with a cervical orthosis, steroids unless contraindicated, and rehabilitation with physical and occupational therapy.
Outcome
Many patients with CCS make spontaneous recovery of motor function while others experience considerable recovery in the first six weeks post injury.
If the underlying cause is edema, recovery may occur relatively soon after an initial phase of motor paralysis or pareses. Leg function usually returns first, followed by bladder control and then arm function. Hand movement and finger dexterity improves last. If the central lesion is caused by hemorrhage or ischemia, then recovery is less likely and the prognosis is more devastating.
The prognosis for CCS in younger patients is favorable. Within a short time, a majority of younger patients recover and regain the ability to ambulate and perform daily living activities. However, in elderly patients the prognosis is not as favorable, with or without surgical intervention.
Become Pain Free | Pain Specialist in Texas
Call Us: (214) 396-3647 | (888) 373-3720 Fax #: (888 )238-9155 | E-mail Us
Regenerative Medicine
Administering undifferentiated stem cells to an injured disc has made adult stem cell regenerative medicine in spine a reality. Adult stem cell regenerative medicine holds the promise of stabilizing or even reversing the degenerative changes associated with aging or following traumatic injury. Current clinical use of stem cells is very limited, in part by the cumbersome approval process. The use of concentrated bone marrow aspirate (BMC) as a “stem cell” preparation is currently the simplest and safest way of utilizing the regenerative potential for mesenchymal stem cells (MSCs) to promote tissue regeneration. In fact, stem cells concentrated from bone marrow have been shown to stimulate the formation of bone, cartilage, ligament and tendon, and dermal tissues.
Conservative treatment options for lumbar and cervical pain associated with discogenic disc disease (Pfirrmann Grades 3-6) are limited. Treatment options include pain medication, steroids, physical therapy, and chiropractic care. Reversal of disc pathology has not been achieved with current available treatment modalities. Failure of these nonoperative treatments may leave surgical intervention as a treatment option.
The goal of utilizing MSCs is to not only potentially provide pain relief from the painful degenerative disc, but to reverse the degenerative process. There are three methods for placing MSCs into the painful nucleus pulposus.
The use of allogeneic mesenchymal precursor cells (MPCs) is currently being evaluated as a part of an FDA Phase I clinical trial. Extracted from donors and expanded in number by tissue culture, this process isolates and grows the stem cells into pure MPCs which are injected into the nucleus pulposus. This technology does not have FDA approval.
Utilizing expanded, autologous MSCs for injection into the painful disc is the second method. Federal regulations require the approval of an Investigational New Drug application supported by prospective, randomized clinical trials for the use of expanded autologous MSCs. The FDA has not approved this technology.
The third method involves autologous point of care therapy. This technology does not require FDA approval. The patient’s own MSCs are directly injected into the nucleus pulposus of the symptomatic degenerated disc(s) using standardized two needle discography technique. This requires fluoroscopic visualization and 2-3cc of MSCs are slowly injected into the symptomatic nucleus pulposus.
There have been no complications associated with the iliac crest aspiration or disc injection. Thus far no patient in the study has undergone spine surgery following treatment. Results obtained with this technique suggest its potential clinical efficacy in the treatment of moderate to severe degenerative disc disease. These results require verification with longer follow-up and randomized prospective studies.
For more information regarding treatment options please visit our websites at http://www.becomepainfree.com
Become Pain Free | Pain Specialist in Texas
Call Us: (214) 396-3647 | (888) 373-3720 Fax #: (888 )238-9155 | E-mail Us
Adult Stem Cell Therapy to Treat Back Pain, Stem Cell, Spine Stem Cells, Stem Cell Treatment
Adult Stem Cell Therapy that doesn’t require FDA approval to treat lumbar and cervical spine conditions resulting from injury or aging, and is also involved with an FDA clinical trial investigating the use of Adult Stem Cells.
These stem cells are autologous – they are taken from an adult patient and returned to that same patient in a concentrated form to the damaged area in a 30-minute procedure. This type of adult stem cell therapy does not require FDA approval to administer.
When it comes to stem cells, there is often a lot of mystique surrounding them. We hear from the media that we can create a human being out of a bundle of cells, which is not necessarily true. We also tend to look at embryonic stem cells as being the only type of stem cell. With these types of embryonic stem cells, one idea is to be able to create a liver or kidney in a Petri dish, which is not controllable or feasible at this point, and the work being done by the BecomePainFree.com medical group.
When we look at stem cell types, we have embryonic stem cells on one hand and adult stem cells on the other. The characteristics of each of these are not like other cells. For instance, a liver cell can divide but it will only ever be a liver cell. These stem cells, both embryonic and adult, can turn into different types of cells. The embryonic stem cells can really turn into any cell type, but adult stem cells are limited as far as the cells they can turn into. This depends upon environment or niche and what they are already programmed to become. A lot of people think there is a lot of promise with embryonic stem cells and there is, although we are not quite there yet.
We are still at the forefront of stem cell technology and embryonic stem cells in particular. With those cells, we do not have the ability to control what types of tissue they turn into. For example, we could be trying to manipulate these cells to turn into kidneys, but they might start to develop as pancreatic cells, which is troublesome. Another key with all stem cells is that they can proliferate quite a bit, usually at a higher rate than just a regular somatic cell. Although this sounds good at first glance, the issue with this, particularly with embryonic stem cells, is we cannot control that division. Hence, these cells can keep going and going without dying. In the normal bodily process, cells are programmed to die after a certain time, but these embryonic stem cells can evade that action and continue dividing, which takes on the characteristic of cancer cells. In some animal studies, an issue that keeps arising is development of tumors in some of these animals. It is difficult to predict if tumors are going to form when using some sort of embryonic stem cell treatment. This is still a scary area through which we are still trying to navigate.
However, the focus of the BecomePainFree.com medical group is on adult mesenchymal stem cells. On the whole, the media does not give a lot of attention to these kinds of stem cells, as using them avoids any kind of ethical or controversial issues. There is a great amount of research being done on adult mesenchymal stem cells, however, because they are very powerful.
First off, we can control what cell type they turn into much more easily. For example, the treatment used by the BecomePainFree.com medical group focuses on Mesenchymal precursor cells (MPC). Mesenchymal means these cells are not going to turn into any kind of blood product such as a red blood cell or white blood cell, although they are derived from bone marrow. The fact that they are precursor cells means these MPCs are only going to differentiate into one of a few cell types. They are either going to become bone cells, i.e., osteoblasts, or chondrocytes, i.e., cartilaginous tissue such that we see in intervertebral discs and joints, etc. All of that really depends on the environment in which we place these adult stem cells where it is well suited to do this. For example, we can inject these MPCs into a bone fracture, and because the cells are surrounded by bone tissue, these cells will receive signals from the surrounding cells that tell them to turn into bone. However, the cells we use will be injected into a disc or joint, and the cells composing the disc and joint tissue will signal the stem cells to develop into similar tissue. Again, there is no chance of any sort of pancreatic cell or nerve cell type spontaneously forming because we are using certain adult stem cell types, which are limited and cannot turn into anything like that. In addition, as the tissue surrounding the disc and joint is relatively avascular, there is not really any worry of these cells migrating through the blood stream to somewhere else in the body and causing any sort of problem. As far as the proliferation issue with embryonic stem cells, we have not seen this issue with adult stem cells in terms of dividing exponentially without ceasing. There is almost a preset limit to how many times these adult stem cells will divide.
Become Pain Free | Pain Specialist in Texas
Call Us: (214) 396-3647 | (888) 373-3720 Fax #: (888 )238-9155 | E-mail Us https://www.becomepainfree.com/